
What is
RAS4?
RAS is our Regulatory Affairs software, a leader in the Brazilian market, operating in 64 countries and 3 continents. It offers complete management for the industry and can be accessed online at any location.
The software has a dossier module in order to meet all the demands of the sector, in addition to the management of regulatory information and KPIs. Therefore, managers can obtain real-time KPIs and alerts to mitigate the regulatory risk of missing important deadlines such as submission of renewals.
Complete Solution
Easy Deployment
Intuitive
Use
Excellent cost-benefit
RAS4
Regulatory Affairs Management
Our customers have a rich set of tools and features, with easy access to information and simplified search. The flexibility of the system also stands out: it is possible to create new fields, without making updates or carrying out customization projects.
• Dashboards configuration;
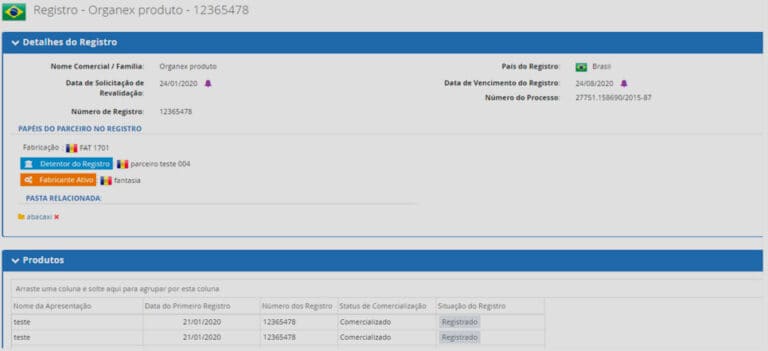
• Submission of deadline alerts for renewal of registers/certificates, submission, and fulfillment of deficient letter;
• Storage of Regulatory Affairs registers/certificates, products, presentations, submissions, partners, and documents;
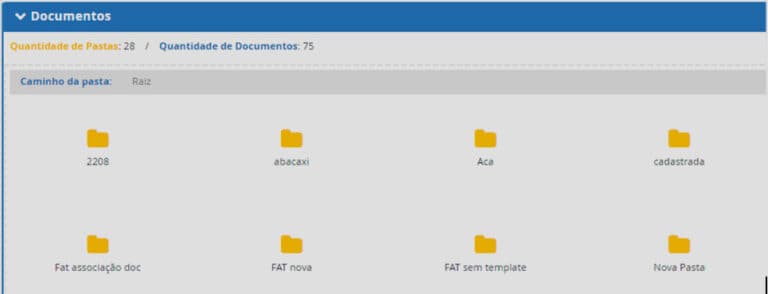
• Organization of regulatory documentation through folders that can be linked to your register/certificates or submission;
• Planning of submission demands and ongoing or planned activities for the team;
• Management of own and third-party factories, including authorizations and certifications;
• Lists setting and new fields in just a few minutes;
• Log and audit of all activities performed, enabling tracking of all details of solution usage and data change history.
You will have a flexible and configurable system that adheres to the needs of the business, which enables you to create and change new fields in a few minutes.
See what you can manage with real-time updated dashboards:
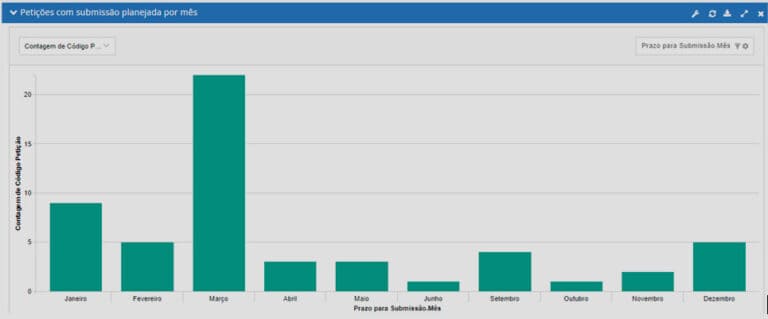
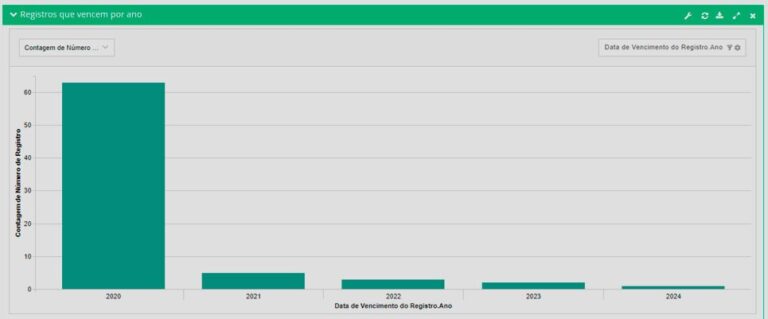

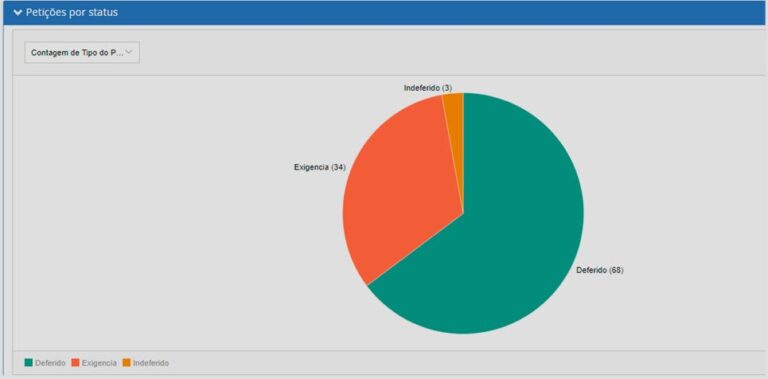
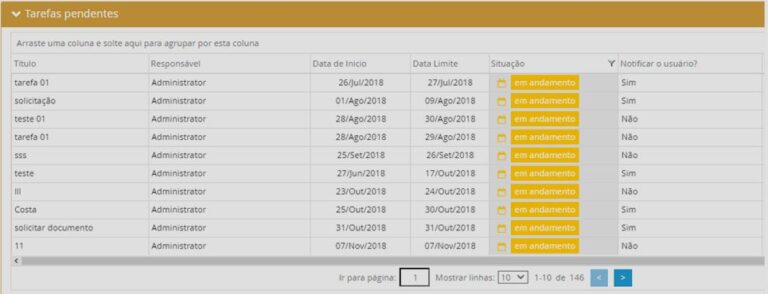
RAS4
Dossier Module
The dossier module is the right tool to automate the process of organizing, managing, and controlling the dossiers. By using it, you can register structures in folders to meet the CTD standard.
• Organization and management of dossiers in CTD and technical dossiers;
• Manage folders in different formats, in a single system;
• Checking the size and extension of the files;
• Dossier documents check;
• Tool in Portuguese and easy access to support;
• Unlimited number of access profiles.
All actions performed are recorded in an audit trail
