Who We Are
Brazilian company with international reach, present in twenty six countries, operating for almost two decades in regulated industries (Pharmaceutical, Cosmetic, Medical Devices, Medical Gases, Food, Cleaning Products, among others), with solid growth and recognized in the market for the quality of its products and services
Reference in Quality system service, especially in activities related to Validations (Systems, Cleaning, and Process), Qualifications (Equipment and Thermal), IT infrastructure Qualification, and Audits in Software and Service providers
Leader in solutions for Regulatory Affairs management with dossier module for compliance with the CTD standard
One of the leading companies in solutions for Post-Marketing monitoring (Pharmacovigilance, Toxicovigilance and Cosmetovigilance) with software in compliance with current standards, including the E2B standard
Our Solutions

RAS4
RAS is our Regulatory Affairs solution, which offers complete management for the industry and can be accessed online from anywhere.

SIS
SIS is our Safety Information Solution. It allows you to record, assess and monitor the occurrence of adverse events related to the use of medication, cosmetics and healthcare products.
Learn more about Organex
Where we are
Since 2006, every choice made by our team in caring about quality, safety, integrity and traceability has brought us closer to this achievement: we have reached 77 countries on 3 continents.
We are in America (North, Central and South), Europe, Asia and Africa. We are Brazilians, and our solutions meet global needs, we do not give up the quality and we seek more and more technology and innovation. We are Organex!
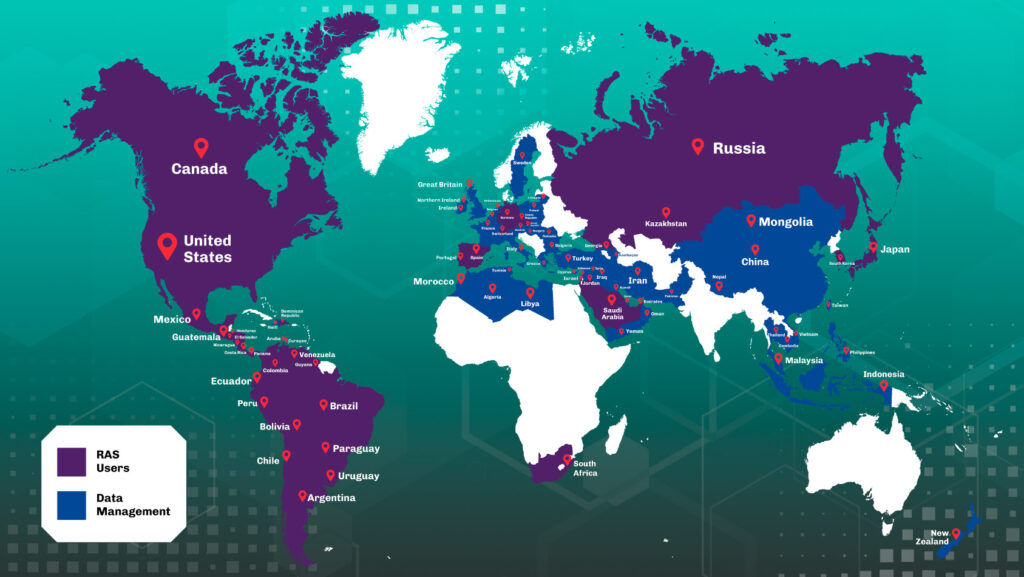
Customers
Testimonials
"I would like to thank the service, support and especially, the partnership for the validation of the UV Chemstation that was made at the end of the year. Even asking for help at the last minute Organex was willing to support us in this important action and it is partners like this that we need and with which we count to continue our journey in the market."
"I have followed several projects from Organex in which both managers and analysts have shown great technical knowledge and commitment to the business. These projects have surely been of great learning and exchange of experiences. I would like to thank the company for the constant support."
"I am extremely grateful for the work done by the entire Organex team. The project management, documentation, and face-to-face meetings were of great value and learning for the entire Hipolabor team. The documentation for validation was already presented for sanitary inspection and was praised not only for the organization of information but also for the quality of work and compliance with standards. New projects are coming!"
"There have been several projects already completed and some still in progress in which we could count on the partnership with Organex. I consider Organex a great partner to help us overcome the daily challenges of such a critical industry as the life sciences. With great professionalism and punctuality, its professionals, technically top-notch in the market, commit themselves to the development of the project and the results have always been very good. We are absolutely sure we will consider Organex for future projects and do not intend to leave this partnership aside."
"The project management was very good, with realistic dates and expectations, strictly following data integrity guidelines."


